Latest News in Cancer
Stay updated with the most recent articles and resources.
Main Topics
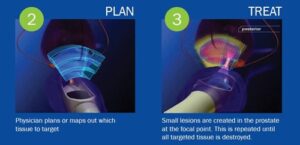
Focal HIFU therapy in prostate cancer
What does local prostate cancer mean? In Liv Hospital Urology Clinic, HIFU is applied as the primary treatment approach in localized prostate cancer, that...
AMC opens up CAR T-Cell therapy center in Seoul
Jan 2023: The Asan Medical Center (AMC) opened the first CAR-T cell treatment facility in the country after the government approved health insurance benefits for Kymriah’s...

Best cancer hospitals in India in 2023
Several notable cancer hospitals in India are known for their superior medical competence and cutting-edge technology. The Tata Memorial Hospital in Mumbai is well-known for...
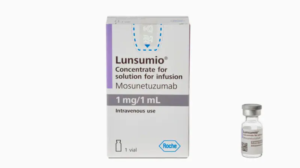
Mosunetuzumab-axgb is granted accelerated approval for relapsed or refractory follicular lymphoma
Jan 2023: Mosunetuzumab-axgb (Lunsumio, Genentech, Inc.), a bispecific CD20-directed CD3 T-cell engager for adult patients with relapsed or refractory follicular lymphoma (FL) following two or...
First adenoviral vector-based gene therapy for high-risk Bacillus Calmette-Guérin unresponsive non-muscle invasive bladder cancer is approved by FDA
Jan 2023: The drug nadofaragene firadenovec-vncg (Adstiladrin, Ferring Pharmaceuticals) has been approved by the Food and Drug Administration for adult patients with high-risk, unresponsive non-muscle...
Adagrasib gets accelerated approval for KRAS G12C-mutated NSCLC
Jan 2023: Adagrasib (Krazati, Mirati Therapeutics, Inc.), a RAS GTPase family inhibitor, was given accelerated approval by the Food and Drug Administration (FDA) for adult...
Atezolizumab is approved by FDA for alveolar soft tissue sarcoma
Dec 2022: Atezolizumab (Tecentriq, Genentech, Inc.) has been approved by the Food and Drug Administration (FDA) for adult and paediatric patients with unresectable or metastatic alveolar...
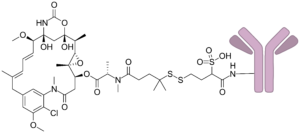
Olutasidenib is approved by FDA for relapsed or refractory acute myeloid leukemia with a susceptible IDH1 mutation
Dec 2022: Olutasidenib (Rezlidhia) capsules were approved by the Food and Drug Administration (FDA) for adult patients with relapsed or resistant acute myeloid leukaemia (AML) who...

New dosing regimen for asparaginase erwinia chrysanthemi (recombinant) is approved by FDA
Dec 2022: A new Monday-Wednesday-Friday dosing schedule for asparaginase erwinia chrysanthemi (recombinant)-rywn has been approved by the Food and Drug Administration (Rylaze, Jazz Pharmaceuticals). Patients...
Mirvetuximab soravtansine-gynx is granted accelerated approval for FRα positive, platinum-resistant epithelial ovarian, fallopian tube, or peritoneal cancer
November 2022: For adult patients who have had one to three prior systemic treatment regimens and have folate receptor alpha (FR) positive, platinum-resistant epithelial ovarian,...
Tremelimumab is approved by FDA in combination with durvalumab and platinum-based chemotherapy for metastatic non-small cell lung cancer
November 2022: The combination of tremelimumab (Imjudo, AstraZeneca Pharmaceuticals), durvalumab (Imfinzi, AstraZeneca Pharmaceuticals), and platinum-based chemotherapy was approved by the Food and Drug Administration for...
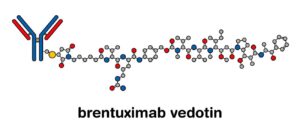
Brentuximab vedotin is approved by FDA in combination with chemotherapy for pediatric patients with classical Hodgkin lymphoma
November 2022: The combination of doxorubicin, vincristine, etoposide, prednisone, and cyclophosphamide with brentuximab vedotin (Adcetris, Seagen, Inc.) has been approved by the Food and Drug...
Need help? Our team is ready to assist you.
We wish a speedy recovery for your loved ones.