Latest News in Cancer
Stay updated with the most recent articles and resources.
Main Topics
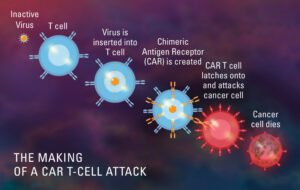
CT103A, CAR T-cell Therapy, has been designated as an orphan drug by the FDA
Feb 2023: The FDA has granted orphan drug status to CT103A, an experimental CAR T-cell therapy being developed by IASO Biotherapeutics and Innovent Biologics to...
CAR T-cell treatment from IASO Biotherapeutics receives new FDA approval
Feb 2023: IASO Biotherapeutics’ investigational CAR T-cell therapy for relapsed or refractory multiple myeloma (RRMM), CT103A, has received fast track and regenerative medicine advanced therapy...
Dostarlimab-gxly is approved by FDA for dMMR endometrial cancer
Feb 2023: Dostarlimab-gxly (Jemperli, GlaxoSmithKline LLC) was given FDA approval to treat adult patients with mismatch repair deficient (dMMR) recurrent or advanced endometrial cancer that...
Sacituzumab govitecan-hziy is approved by FDA for HR-positive breast cancer
Feb 2023: The Food and Drug Administration (FDA) has approved sacituzumab govitecan-hziy (Trodelvy, Gilead Sciences, Inc.) for people with hormone receptor (HR)-positive, HER2-negative (IHC 0,...
Elacestrant is approved by FDA for ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer
In February 2023, the Food and Drug Administration (FDA) approved elacestrant (Orserdu, Stemline Therapeutics, Inc.) for women or men over 50 who have advanced or...

Accelerated approval is granted by FDA to pirtobrutinib for relapsed or refractory mantle cell lymphoma
Feb 2023: Accelerated approval is granted by FDA to pirtobrutinib (Jaypirca, Eli Lilly and Company) for relapsed or refractory mantle cell lymphoma. In BRUIN (NCT03740529),...
Pembrolizumab is approved by FDA as adjuvant treatment for non-small cell lung cancer
Feb 2023: For stage IB (T2a 4 cm), stage II, or stage IIIA non-small cell lung cancer, the Food and Drug Administration (FDA) approved pembrolizumab...
Zanubrutinib is approved by FDA for chronic lymphocytic leukemia or small lymphocytic lymphoma
Feb 2023: Zanubrutinib (Brukinsa, BeiGene USA, Inc.) is approved by FDA for chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). SEQUOIA was used to...

Accelerated approval is granted by FDA to tucatinib with trastuzumab for colorectal cancer
In February 2023, the Food and Drug Administration (FDA) sped up the approval of tucatinib (Tukysa, Seagen Inc.) and trastuzumab for the treatment of RAS...
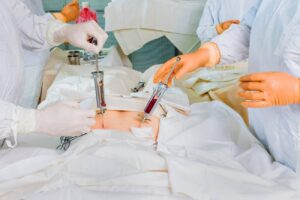
Bone marrow transplant in Iran
Bone marrow transplant is one of the services of Cancerfax, which is provided by the best surgeons, along with accommodation, a translator, a companion nurse,...
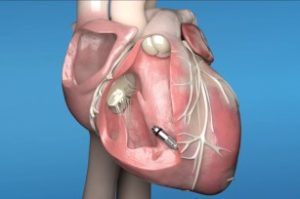
Sheba medical center became the first Israeli facility to receive permission from Medtronic International to train medical professionals on MICRA pacemaker implants
RAMAT GAN, Israel – June, 2022 – Sheba Medical Center, Israel’s largest medical center and a Newsweek top-10 ranked world’s best hospital for the last...

Utilization of wearable telehealth technology for remote patient monitoring is validated by new Sheba medical center study
July 2022: Peer-reviewed study analyzes usability of a wearable RPM device, which detected early warning of risk for ABCNO deterioration in 75% of patients, 38 hours...
Need help? Our team is ready to assist you.
We wish a speedy recovery for your loved ones.