Latest News in Cancer
Stay updated with the most recent articles and resources.
Main Topics
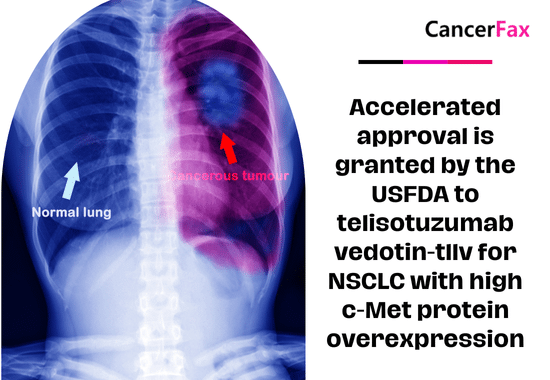
Accelerated approval is granted by the USFDA to telisotuzumab vedotin-tllv for NSCLC with high c-Met protein overexpression
On May 14, 2025, the FDA granted accelerated approval to Emrelis (telisotuzumab vedotin) for treating adults with high c-Met protein overexpressing non-small cell lung cancer...

Belzutifan is approved by the USFDA for pheochromocytoma or paraganglioma
On May 14, 2025, the FDA approved belzutifan (Welireg) for treating adults and children aged 12 and older with locally advanced, unresectable, or metastatic pheochromocytoma...

Accelerated approval is granted by the USFDA to the combination of avutometinib and defactinib for KRAS-mutated recurrent low-grade serous ovarian cancer
The FDA has granted accelerated approval to the combination therapy of avutometinib and defactinib for treating KRAS-mutated recurrent low-grade serous ovarian cancer. This decision is...
Chimerix’s Dordaviprone Receives FDA Priority Review for Recurrent H3 K27M-Mutant Diffuse Glioma
Chimerix’s drug Dordaviprone has received FDA priority review for treating recurrent H3 K27M-mutant diffuse glioma. With a PDUFA date of August 18, 2025, it could...
Penpulimab-kcqx is approved by the USFDA for non-keratinizing nasopharyngeal carcinoma
Penpulimab-kcqx, developed by Akeso Biopharma, has received FDA approval for treating recurrent or metastatic non-keratinizing nasopharyngeal carcinoma. This PD-1 inhibitor marks a major advancement in...

Dr. Zhang Wei: Leading Innovation in Epigenetic Therapy for Diffuse Midline Glioma at Tsinghua University
Dr. Zhang Wei of Tsinghua University leads pioneering research in epigenetic-based immunotherapy for Diffuse Midline Glioma (DMG). His work integrates advanced diagnostics, precision medicine, and...
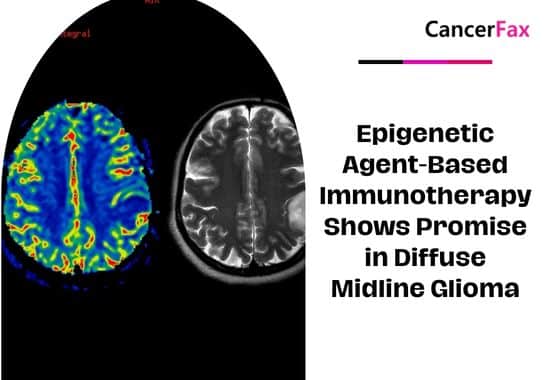
Epigenetic Agent-Based Immunotherapy Shows Promise in Diffuse Midline Glioma
A groundbreaking case study reveals that epigenetic agent-based immunotherapy significantly regressed tumors in a patient with diffuse midline glioma (DMG), extending survival to 20 months....

Breakthrough in Cancer Treatment: FDA Approves Drug from Small Enterprise in Tianjin, China
Introduction In a stunning surprise that has stunned the international biotech industry, a tiny and micro business in Tianjin, China, has come up with an...

ONC201 Capsules: A New Hope for Brain Glioma Patients in China
Gliomas Introduction Brain gliomas, especially aggressive types such as glioblastoma multiforme (GBM), have long been extremely challenging to treat because of their insensitivity to traditional...

Medical Tourism in China: Best Hospitals, Costs & Treatments (2025 Guide)
China has become a leading destination for medical tourism, providing high-end treatment at 30-70% lower prices compared to Western nations. Ranging from state-of-the-art cancer treatments...
4th Generation CAR T Cell Therapy with IL-15: A new era in cancer treatment
4th generation CAR T-cell therapy with IL-15 represents a groundbreaking advancement in cancer immunotherapy. By incorporating IL-15, these engineered T cells exhibit enhanced persistence, potency,...
Nivolumab with ipilimumab is approved by the USFDA for unresectable or metastatic hepatocellular carcinoma
On April 11, 2025, the Food and Drug Administration sanctioned nivolumab (Opdivo, Bristol Myers Squibb Company) in conjunction with ipilimumab (Yervoy, Bristol Myers Squibb Company)...
Need help? Our team is ready to assist you.
We wish a speedy recovery for your loved ones.