Latest News in Cancer
Stay updated with the most recent articles and resources.
Main Topics

Advancing Bardet-Biedl Syndrome Treatment in China: Gene Therapy and Clinical Innovations at NCMC, Shanghai
Certainly! Here's the **70-word excerpt** for the **previous blog post** on **Bardet-Biedl Syndrome**: --- Bardet-Biedl Syndrome (BBS) is a rare genetic disorder with complex symptoms....
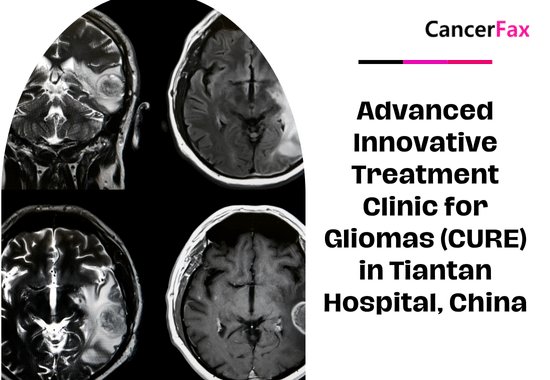
Advanced Innovative Treatment Clinic for Gliomas (CURE) in Tiantan Hospital, China
Beijing Tiantan Hospital stands at the forefront of glioma treatment in China, offering advanced therapies and participating in pioneering clinical trials. With a focus on...

Review and Prospect of Brain Glioma Treatment in China
China is advancing in brain glioma treatment through updated clinical guidelines and innovative therapies like immunotherapy and CAR-T cell therapy. Despite challenges such as low...

Shen Baiyong’s Pancreatic Cancer Innovation Studio in Shanghai, China
Dr. Shen Baiyong’s Pancreatic Cancer Innovation Studio at Ruijin Hospital, Shanghai, is pioneering advancements in early detection, precision treatment, and drug resistance in pancreatic cancer....
Safety and Tolerability of VGB-R04 in Patients With Hemophilia B
This Chinese trial is evaluating VGB-R04, a new gene therapy for Hemophilia B that uses a single AAV infusion to restore Factor IX activity. The...
A Study to Evaluate the Safety and Efficacy of ZS801 in Adult Hemophilia B Patients
A phase I/II gene therapy trial in China is testing ZS801, an AAV vector delivering the human Factor IX gene for adults with Hemophilia B....

A Clinical Study Evaluating the Safety and Efficacy of SKG0201 Injection in Patients With Spinal Muscular Atrophy Type 1
A groundbreaking gene therapy trial in China is testing SKG0201, a single-dose rAAV-based treatment for Spinal Muscular Atrophy Type 1 (SMA 1). Conducted by Xinhua...
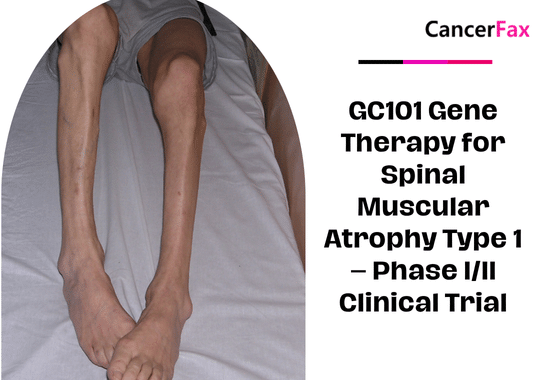
GC101 Gene Therapy for Spinal Muscular Atrophy Type 1 – Phase I/II Clinical Trial
This Phase I/II trial tests GC101 gene therapy in infants with Type 1 Spinal Muscular Atrophy. Delivered intrathecally as a one-time treatment, GC101 aims to...

Evaluation of Safety and Efficacy of Gene Therapy Drug in the Treatment of Spinal Muscular Atrophy (SMA) Type 2 Patients
This Phase I/II trial investigates GC101, a gene therapy using AAV9 vectors to treat children with Spinal Muscular Atrophy Type 2. Administered as a one-time...
Evaluation of Safety and Efficacy of Gene Therapy Drug in the Treatment of Spinal Muscular Atrophy (SMA) Type 3 Patients
This clinical trial investigates the safety and efficacy of GC101 gene therapy in treating Type 3 Spinal Muscular Atrophy (SMA). The study involves a single...

Safety and Efficacy Evaluation of GC101 Gene Therapy Via Intrathecal (IT) Injection in the Treatment of Patients With Type 2 Spinal Muscular Atrophy (SMA) – Phase III
This Phase III clinical trial evaluates GC101 gene therapy for children aged 2–12 with Type 2 Spinal Muscular Atrophy (SMA). Sponsored by GeneCradle Inc., the...
Retifanlimab-dlwr with carboplatin and paclitaxel is approved by the USFDA and as a single agent for squamous cell carcinoma of the anal canal
On May 15, 2025, the FDA approved Zynyz (retifanlimab-dlwr) in combination with carboplatin and paclitaxel, and as a single agent, for treating adults with inoperable...
Need help? Our team is ready to assist you.
We wish a speedy recovery for your loved ones.