CAR T-Cell far don ciwon huhu mara ƙanƙanta
Hanyar juyin juya hali a cikin maganin ciwon huhu.
Kuna son yin rajista a cikin wannan ci gaban maganin ciwon daji?
March 2024 : CAR T-cell therapy is a potential cancer treatment strategy, notably in hematological malignancies. However, its effectiveness in solid tumors, such as lung cancer, is limited due to the tumor microenvironment’s immunosuppressive nature. Researchers are developing next-generation CAR T cells to boost their infiltration, survival, and persistence inside malignancies. Clinical trials are underway to assess the safety and efficacy of CAR T-cell treatments in lung cancer, with some yielding promising results. Antigen escape, immunological barriers, and on-target off-tumor damage are among the challenges in CAR T-cell treatment for lung cancer. Engineering CAR constructs, altering the tumo microenvironment, and employing off-the-shelf CAR T cells are some strategies for addressing these problems.
One of the leading hospitals in China has successfully conducted trials of CAR T-Cell therapy in cututtukan daji na kansa marasa kansar patients. CAR T-Cell on all these cancers is applicable for patients after some lines of treatment such as surgery, chemotherapy and radiotherapy but relapsed.
Of all malignancies, lung cancer has the greatest incidence and fatality rates worldwide. A growing variety of immunotherapeutic medicines, particularly those that target monoclonal antibodies, have been employed in the clinical treatment of malignancy in the current immunotherapy period, although it still has numerous drawbacks. In addition to being utilised successfully against haematological cancers, chimeric antigen receptor-modified T (CAR-T) cells have also created new opportunities for the immunotherapy of solid tumours, such as lung cancer. The lack of appropriate tumor-specific antigens, an immunosuppressive tumour microenvironment, a low level of CAR-T cell penetration into tumour tissues, together with off-target effects, etc. make it difficult to target lung cancer-specific antigens with modified CAR-T cells. Meanwhile, due to numerous difficulties such as tumor lysis syndrome, neurotoxicity syndrome, and cytokine release syndrome, the clinical usage of CAR-T cells is still restricted. With the goal of offering fresh perspectives and methods for pre-clinical studies and clinical trials of CAR-T cell therapy for lung cancer, we outline the fundamental structure and generation characteristics of CAR-T cells in this review, summarise the typical tumor-associated antigens, and highlight the current challenges.
Tsarin MOtoci
Tun daga farkonsa, amfani da CARs a cikin maganin T-cell ya wuce ta al'ummomi guda huɗu, waɗanda duk sun dogara ne akan wuraren siginar ciki na CAR. Ƙarni na farko na CARs suna da raunin aiki da ɗan gajeren lokacin rayuwa saboda kawai sun ƙunshi siginar gane antigen. Wurin watsa siginar na ƙarni na biyu da na uku CARs, bi da bi, ya ƙunshi ƙwayoyin cuta guda ɗaya da biyu. An yi waɗannan canje-canjen don ƙara yawan rayuwa T cell, cytotoxicity, da yaduwa. An inganta ƙwayoyin haɗin gwiwar motsa jiki a cikin CARs, wanda ya inganta aikin CAR-T. 4-1BB ko CD28 sune yanki na haɗin gwiwa na ƙarni na biyu waɗanda galibi ana amfani da su akai-akai. Bugu da ƙari, cytotoxicity, samar da cytokine, da kunna T cell duk an nuna an inganta su ta hanyar DNAX mai kunna furotin 10 (DAP10). Bisa layukan salula marasa ƙananan ƙwayoyin huhu (NSCLC), jinkirta ci gaban ciwon huhu na farko da kuma ƙara yawan aikin rigakafin ƙwayar cuta an nuna su a cikin vivo nau'in dabba na ƙwayar cutar kansar huhu na ɗan adam xenotransplantation. An ƙara cytokines masu kumburi da haɗin gwiwa a cikin ƙirar CAR-T na ƙarni na huɗu don taimakawa T-cell ɗin kutsawa kuma su wuce abubuwan hana TME masu ƙiyayya.
Ƙwaƙwalwar haɓakawa da ingantaccen ƙwayar ƙwayar cuta na ƙwayoyin CAR-T an nuna su don haɓakawa ta hanyar haɓaka tsarin ƙirar ƙwayoyin cuta ban da na'urorin watsa siginar ciki. A cewar Qin et al., guntu mai jujjuya sarkar guda ɗaya (scFv), wanda ke ɗaure da ƙarfafa haɓakawa, ƙaura, da mamaye gungu na bambance-bambancen 4 (CD4) + CAR-T, an sanya shi mafi sauƙi ta ƙari. na tsarin hinge. Kodayake sel CAR-T na ƙarni na biyu suna ci gaba da kasancewa daidaitaccen hanyar aikace-aikacen warkewa, tsarin gine-ginen CARs koyaushe yana inganta kuma yana da mahimmanci ga tasirin CAR. T na
CAR T-Cell far a cikin ciwon huhu da kuma antigen da aka yi niyya
Lokacin da aka bayyana abin da ake nufi-antigen a kan ƙwayoyin cutar kansa ko kuma ya wuce gona da iri akan duk ko yawancin ƙwayoyin cutar kansar huhu idan aka kwatanta da sel na al'ada, wannan shine mafi kyawun manufa don maganin cell CAR-T. Ko da yake an sami adadin adadin antigens masu alaƙa da ƙari (TAA) a cikin cututtukan da ba ƙananan ƙwayoyin cuta ba (NSCLCs), kaɗan ne kawai daga cikin waɗannan antigens an yi niyya musamman ta sel CAR-T (8). Bugu da ƙari, wasu daga cikin waɗannan abubuwan-antigens kuma ana bayyana su da rauni a cikin kyallen jikin lafiya, suna ba wa wasu ƙwayoyin CAR-T ikon kai hari ga ƙwayoyin lafiya.
Epidermal girma factor receptor (EGFR), ɗan adam epidermal girma factor receptor 2 (HER2), mesothelin (MSLN), prostate stem cell antigen (PSCA), mucin 1 (MUC1), carcinoembryonic antigen (CEA), tyrosine kinase-kamar marayu receptor. ROR1), shirye-shiryen mutuwa ligand 1 (PD-L1), da CD80/CD86 suna daga cikin makasudin da ake nazari a halin yanzu don CAR.
Majinyacin ciwon huhu yana jurewa CAR T-Cell far
Nuwamba Nuwamba na 2009, mai haƙuri ya sami tarin huhu na hagu kuma ya yi aikin tiyata mai rauni na hagu. Pathology: huhu adenocarcinoma;
Daga Jan 2013 zuwa Jan 2017, metastases na kwakwalwa guda uku sun faru, kuma an ba da tiyata da maganin radiation a jere tare da rashin kulawa;
Daga Maris 2017 zuwa Satumba 2017, don matakan kwakwalwa, mesoCAR-αPD1 kwayoyin da ke bayyana PD-1 antibody an ba su kwasa-kwasan 6 na magani. Bayan jiyya, an kimanta PR kuma ciwace-ciwacen ya ragu sosai da ƙananan ragowar.
CAR T-Cell far a China
CAR-T Cell therapy a China yana girma cikin sauri sosai. Sakamako na CAR T-cell far a kasar Sin kuma adadin maganin gabaɗaya yana cikin mafi kyau a duniya a halin yanzu. Akwai gwaje-gwajen asibiti sama da 300 da ke faruwa a ciki Sin don CAR T Cell therapy. Kasar Sin tana cikin kasashe na farko da suka ba da maganin CAR T Cell bayan Amurka & UK. Dangane da adadin gwajin asibiti na CAR-T, China ce ta biyu bayan Amurka, tana yin rijista kusan kashi 33% na gwaji a duk duniya. Yawan hanyoyin kwantar da hankali na CAR T-cell a cikin ci gaban asibiti ya yi roka a cikin 'yan shekarun nan. A halin yanzu, a kasar Sin, ana gudanar da gwaje-gwaje na asibiti sama da 300 a cikin cututtukan cututtukan jini da kuma ciwace-ciwace.
China’s extensive explorations and breakthroughs in the search of novel target antigens, optimization of CAR structure, cocktail CAR-T therapy, combination therapy, and extension of CAR-T cell applications, imply that we are currently on the verge of a revolution in CAR-T therapy. US FDA has approved CAR T Ciwon ƙwayar cuta for relapsed B Acute lymphoblastic leukemia, lymphoma & multiple myeloma. Sin kwanan nan ya amince da CAR T-Cell far don wasu m ciwon daji. Mai yiwuwa majinyata daga ko'ina cikin duniya za su ci gajiyar wannan ci gaban.
Kuna so karanta: CAR T-Cell far a China
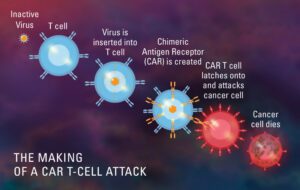
Menene CAR T-Cell farfadowa (Masu karɓa na antim Chimeric)?
CAR T-Cell therapy wani nau'i ne na rigakafi wanda ke amfani da ƙwayoyin T-cell na musamman waɗanda ke cikin tsarin garkuwar jikin mu don yaƙar ciwon daji. Ana tattara samfurin kwayoyin T marasa lafiya daga jini, sannan an gyara shi don samar da sifofi na musamman da ake kira chimeric antigen receptors (CAR) a saman su. Lokacin da aka sake shigar da waɗannan ƙwayoyin CAR da aka gyara a cikin majiyyaci, waɗannan sabbin ƙwayoyin suna kai hari kan takamaiman antigen kuma suna kashe ƙwayoyin ƙari.
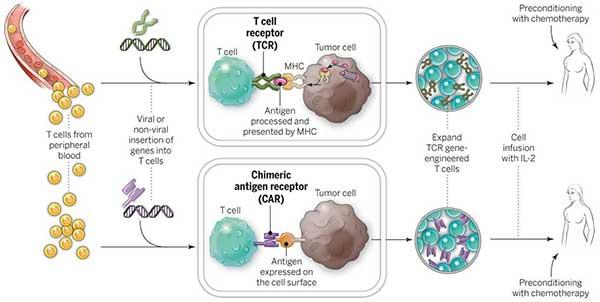
Ta yaya maganin C-T-Cell ke aiki?
CAR T-cell far yana ɗaukar taimako daga tsarin garkuwar jiki don kai hari da kashe ƙwayoyin cutar kansa. Ana yin hakan ne ta hanyar cire wasu takamaiman sel daga jinin majiyyaci, gyara su a cikin dakin gwaje-gwaje da sake yin allura a cikin majiyyaci. CAR T-cell far ya haifar da sakamako mai ƙarfafawa sosai a cikin lymphoma na Non-Hodgkin kuma FDA ta amince da shi.
Wanene 'yan takarar da suka dace don CAR T-Cell far?
A halin yanzu FDA ta amince da CAR T-Cell therapy don wasu nau'ikan lymphoma marasa Hodgkin na muni da kuma sake dawowa da cutar sankarar lymphoblastic mai tsanani. Mara lafiya yana buƙatar aika cikakkun rahotannin likita don tabbatar da amfani da CAR T-Cell far don maganin sa.
Ka'idojin hadawa don C-T-cell far:
1. Marasa lafiya tare da CD19+ B-cell Lymphoma (Aƙalla 2 kafin hadewar chemotherapy)
2. Yayi shekaru 3 zuwa 75
3. ECOG ci ≤2
4. Matan da ke da damar haihuwa dole ne a yi gwajin ciki na fitsari kuma an tabbatar da rashin kyau kafin magani. Duk marasa lafiya sun yarda su yi amfani da ingantattun hanyoyin hana haihuwa a lokacin gwajin har zuwa bin diddigin na ƙarshe.
Ka'idojin keɓancewa don C-T-cell far:
1. Hawan jini a cikin kwakwalwa ko suma
2. Rashin numfashi
3. Yada yaduwar jijiyoyin cikin jini
4. Hematosepsis ko Ciwo mai aiki mara ƙarfi
5. Ciwon suga mara tsari
Fa'idodi na CAR T-Cell far
- > Motocin 5000 CAR T waɗanda kwararrun likitoci suka yi.
- Asibitoci a China sun haɓaka ƙarin nau'ikan CAR T Cell da suka haɗa da CD19 & CD 22 sannan kowace ƙasa a duniya.
- China tana gudanar da gwaji na asibiti sama da 300 a kan maganin CAR T Cell. Fiye da kowace ƙasa a duniya.
- Tasirin asibiti na CAR T Cell yayi kama da na Amurka ko wata ƙasa kuma wani lokacin mafi kyau.
Tsarin magani don CAR T-Cell far
- Cikakken kimantawa na mai haƙuri
- T-cell tarin daga jiki
- T-sel ana yin aikin injiniya a cikin lab
- T-Cells da aka ƙera halittarsu an ninka su ta hanyar amfani da su a cikin dakin bincike. Wadannan kwayoyin sun daskarewa sannan kuma a tura su zuwa cibiyoyin kulawa.
- Kafin infusing, mai haƙuri za a iya ba da ilimin kimiya don ciwon daji. Wannan yana taimaka aikin warkewa ta hanya mafi kyau.
- Ba da daɗewa ba bayan an gama shigar da ƙwayoyin cuta na CAR ta hanyar aiwatarwa wanda yayi kama da shigar jini.
- Akwai watanni 2-3 na lokacin dawowa ga mai haƙuri.
Tsarin lokaci don CAR T-Cell far
1. Jarrabawa & Gwaji: sati guda
2. Pre-treatment & T-Cell Collection: sati guda
3. T-Cell shiri & dawowa: makonni biyu-uku
4. Nazarin Inganci na 1: makonni uku
5. Nazarin inganci 2: makonni uku.
Sakamakon sakamako na CAR T-Cell far
Sakamakon illa na CAR T-cell far sun haɗa da:
- Ciwon saki na Cytokine
A wasu lokuta, marasa lafiya na iya samun alamun mura kamar zazzabi, sanyi, ciwon kai, tashin zuciya, amai, rashin kwanciyar hankali, da tsoka ko ciwon gabobi. Hakanan yana iya haifar da ƙarancin hawan jini, wahalar numfashi, da saurin bugun zuciya. Wadannan illolin suna faruwa ne saboda sakin cytokines ta ƙwayoyin rigakafi yayin maganin CAR T-cell. Waɗannan alamun yawanci suna da sauƙi, amma suna iya zama mai tsanani kuma suna barazanar rayuwa a wasu marasa lafiya. - Ayyukan Neurology
Abubuwan da ke tattare da jijiyoyin jiki na iya faruwa kuma suna iya zama mai tsanani ga wasu marasa lafiya. Irin waɗannan abubuwan sun haɗa da encephalopathy (rauni na kwakwalwa da rashin aiki), rikicewa, wahalar magana, tashin hankali, kamuwa, bacci, yanayin canjin da aka samu da rashin daidaito. - Neutropaenia da Anemia
Wasu marasa lafiya na iya haɓaka neutropenia ko ƙananan fararen ƙwayoyin cuta. Hakazalika, anemia ko ƙananan ƙwayoyin jini na iya faruwa saboda wannan maganin.
.
Abin farin ciki, yawancin waɗannan illolin yawanci suna warwarewa da kansu ko ana iya sarrafa su tare da amfani da magunguna.
Yaya tasirin CAR T-Cell yake tasiri?
CAR T-cell far don maganin lymphoma da sauran cututtukan jini sun nuna kyakkyawan sakamako. Tun lokacin da aka yi maganin C-T-cell, yawancin marasa lafiya waɗanda suka sake dawowar ciwace-ciwacen jini suna da sakamako mai gamsarwa kuma babu shaidar cutar kansa. Hakanan ya taimaka wajen gyara marasa lafiyar wadanda a baya suka kasa amsa mafi yawan magungunan gargajiya na gargajiya.
Koyaya, ana buƙatar nazarin lokaci mai tsawo don yawan marasa lafiya don tabbatar da ingancin wannan magani. Experananan gwaje-gwajen za su iya taimakawa wajen gano yiwuwar tasirin illa da hanyoyin da suka dace don magance su.
Nawa ne kudin CAR T-Cell?
Kasar Sin ita ce jagorar duniya na CAR-T cell far & BMT. Ya zuwa yanzu akwai gwaje-gwajen asibiti sama da 300 na CAR-T da ke ci gaba. Maganin CAR-T na kasar Sin shine mafi kasafin kudi a duk duniya. Saboda shirye-shiryen salula na CAR-T kyauta ne yanzu! Marasa lafiya dole ne su biya kuɗin magani & sabis kawai. Jimlar kuɗin magani zai kasance kusan $60,000 - $80,000.
Har ila yau karanta wannan: CAR T Cell far in India
Ta yaya zan iya shan maganin CAR T-Cell a China?
Mara lafiya na iya kiran +91 96 1588 1588 ko imel zuwa cancerfax@gmail.com tare da cikakkun bayanai na haƙuri da rahotannin likita kuma za mu shirya ra'ayi na biyu, tsarin jiyya da kimanta kashe kuɗi.